Korean AI-Medical startup Lunit has entered into a partnership with Philips. for its diagnostic X-Ray Solution. The company announced it during the European Congress of Radiology (ECR) virtual event (March 3-7, 2021). The collaboration will make Lunit’s AI software accessible to users of Philips’ diagnostic X-ray solutions.
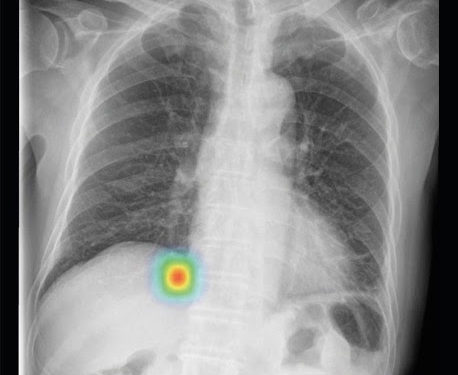
Lunit’s INSIGHT CXR chest detection suite accurately detects 10 of the most common findings in a chest X-ray. The announcement is the latest extension of Philips’ AI portfolio in precision diagnosis, which leverages this collaboration to achieve better patient outcomes, improve the experience of patients and staff, and lower the cost of care.
“By partnering with Philips, one of the biggest medical device companies globally, our AI will be available to its significant global installed base,” said Brandon Suh, CEO of Lunit. “With the start of this partnership, we look forward to further expanding our collaboration to make data-driven medicine the new standard of care. Lunit will continue to build upon its current AI offering, making it better and better with time, and will continue to deliver best-in-class AI.”
“For most patients, X-ray is the first diagnostic imaging step on their path to a definitive diagnosis,” said Daan van Manen, General Manager for Diagnostic X-ray at Philips. “Radiology departments and their technologists are continually under pressure. They face high patient volumes, and every improvement in workflow can make a big impact. The high performance of Lunit’s AI has been validated in major clinical journals to improve accuracy and efficiency in workflow. Our partnership with Lunit to incorporate their diagnostic AI into our X-ray platform combines with a host of smart workflow features in the Philips Eleva user interface, our common platform across our digital radiography systems that enables a smooth and efficient, patient-focused workflow.”
Lunit’s AI software, Lunit INSIGHT CXR, is designed to provide accurate and instant analysis of chest X-ray images by mapping the location of the findings and displaying the scored calculation of its actual existence. It accurately detects 10 of the most common findings in a chest X-ray, prioritizes cases with abnormality scores, thereby allows fast triage of normal cases and enables radiologists to focus on reading abnormal cases. The algorithm performs at 97-99% accuracy rate, with its performance validated in major publications such as Radiology, JAMA Network Open, etc.
Since its foundation in 2013, Lunit has been actively involved in partnerships with global major vendors and expanding its global presence in the X-ray market. Lunit INSIGHT CXR is CE marked and clinically available in Europe, Middle East, Latin America, South East Asia, Australia, and New Zealand. It is expecting its FDA clearance within 2021.